
The values of effective and spontaneous magnetic moment, µeff and µs,... | Download Scientific Diagram
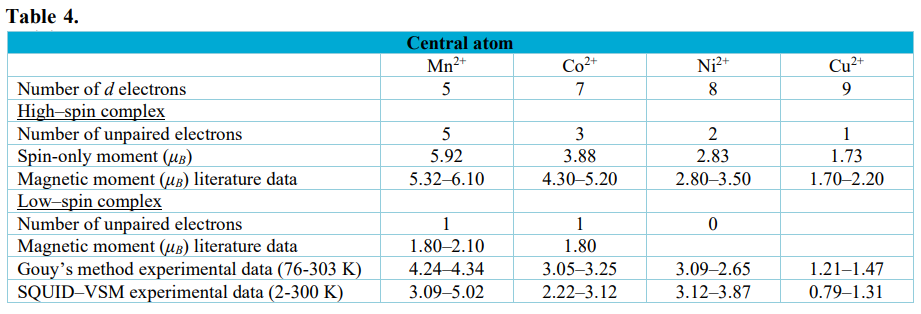
Complexes of Mn(II), Co(II), Ni(II), Cu(II) and Zn(II) with ligand formed by condensation reaction of isatin with glutamic acid

The effective magnetic moments of Co2+ and Co3+ in SrTiO3 investigated by temperature-dependent magnetic susceptibility - ScienceDirect

Table 4 from Silylamide complexes of chromium(II), manganese(II), and cobalt(II) bearing the ligands N(SiHMe2)2 and N(SiPhMe2)2. | Semantic Scholar

The effective magnetic moments of Co2+ and Co3+ in SrTiO3 investigated by temperature-dependent magnetic susceptibility - ScienceDirect

SciELO - Brasil - Magnetic, thermal and spectral properties of Ni(II) 2,3- , 3,5- and 2,6-dimethoxybenzoates Magnetic, thermal and spectral properties of Ni(II) 2,3- , 3,5- and 2,6-dimethoxybenzoates
The magnetic moments of two bar magnets of same size are in the ratio 1:2 when they are placed one over the other with theirsimilar poles together then their period of oscillation

Table 2 from Paramagnetism of aqueous actinide cations. Part I: Perchloric acid media. | Semantic Scholar
Calculate the spin only magnetic moment of La^3+. - Sarthaks eConnect | Largest Online Education Community

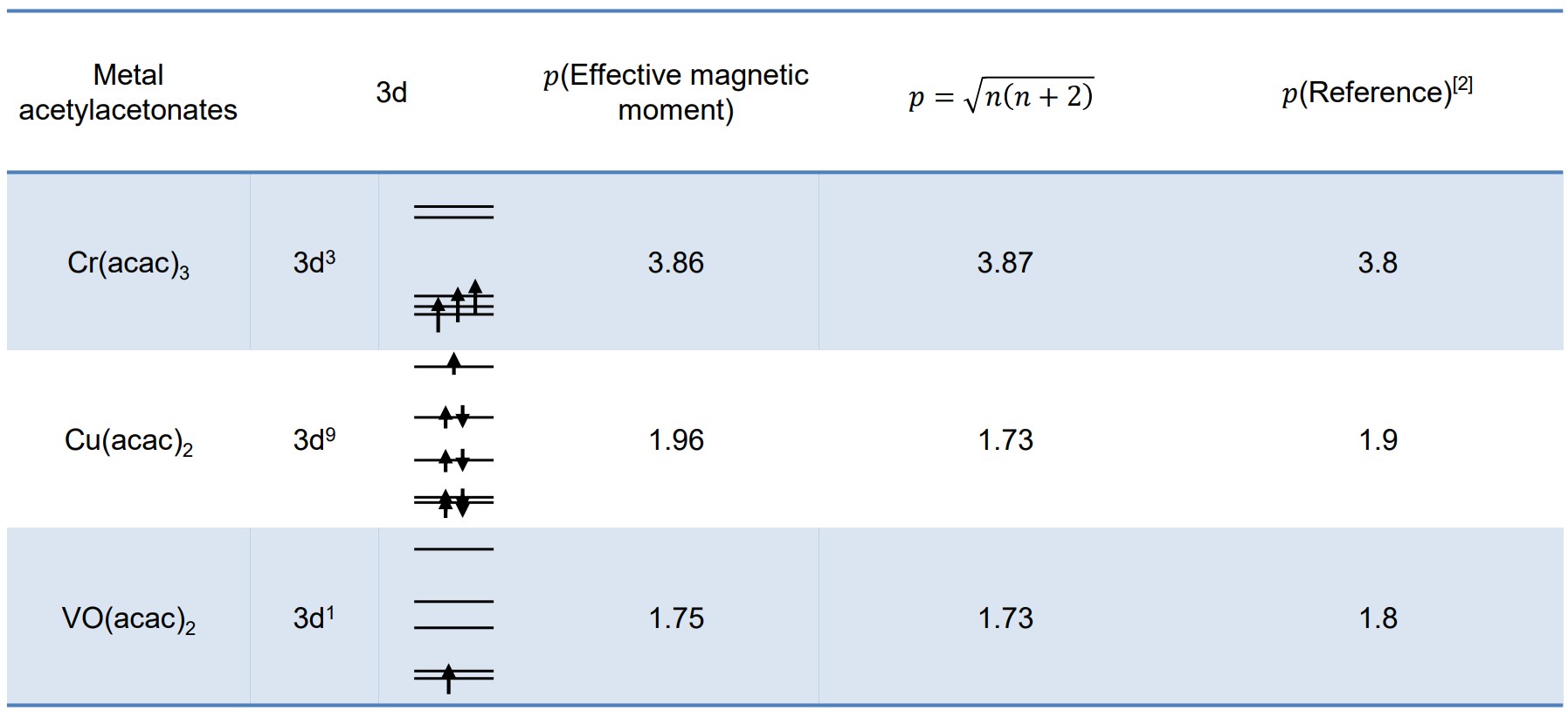




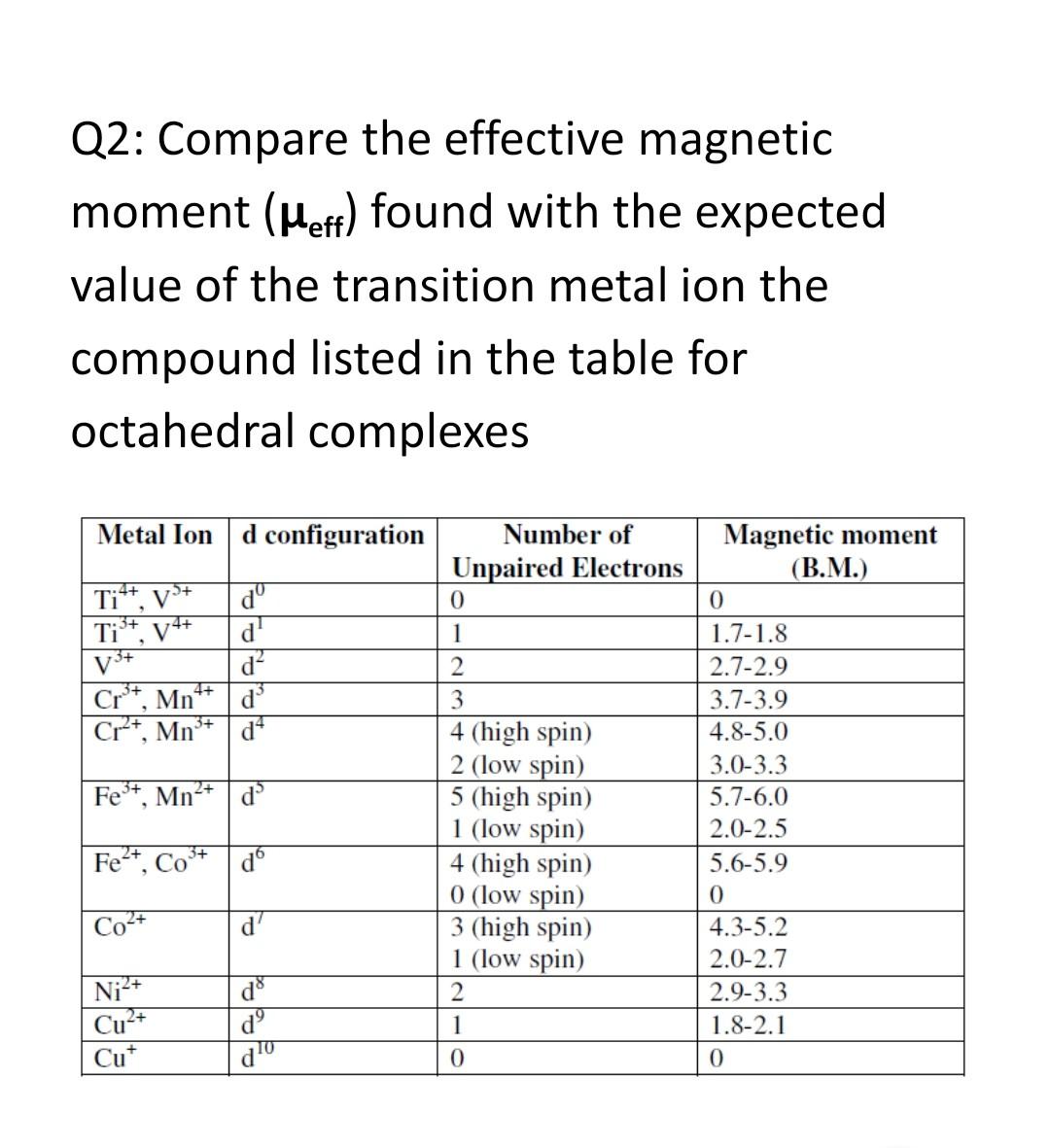
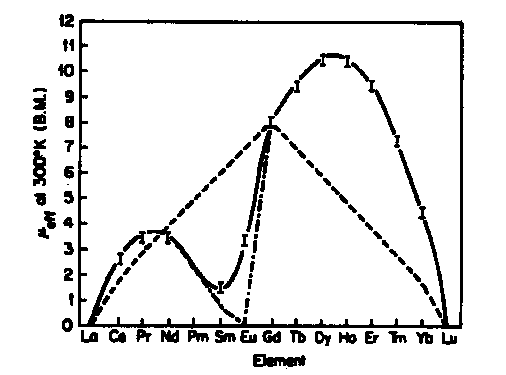






![The effective magnetic moment (μeff) [9] of the four lanthanide ion... | Download Table The effective magnetic moment (μeff) [9] of the four lanthanide ion... | Download Table](https://www.researchgate.net/publication/51852564/figure/fig17/AS:341129435336718@1458343003367/The-effective-magnetic-moment-meff-9-of-the-four-lanthanide-ion-complexes-and-the.png)